|
kansas state university associate professor gupreet singh has patented a liquid polymer composed of silicon, boron, carbon, nitrogen, and hydrogen that turns into a ceramic when heated and has thermal, optical, and electronic properties, according to an article on the school’s website.
the new material turns from liquid to solid when heated. (kansas state university)
the article explained, “the engineers developed the clear polymer that looks like water and has the same density and viscosity as water, unlike some other silicon- and boron-containing polymers.”
it stays a liquid at room temperature, but transforms into a black, glass-like ceramic when heated. among its beneficial qualities, the polymer is low density and can be used to create lightweight ceramics; it is scalable and cane be produced in grams or kilograms; it can withstand temperatures as high as 1,700°c; when heated to 50-100°c, it becomes a gel that can be pulled into strings to create ceramic mesh; it can be poured into molds for complex shapes; and can be sprayed in liquid form and heated to create ceramic coatings.
the article also noted that the polymer can be combined with carbon nanotubes to form a material that absorbs all light without being damages and can withstand thermal levels of as much as 15,000 w/cm2.
the polymer can be tuned to be either an insulator or semiconductor and the presence of silicon and graphene-like carbon in the ceramic can improve electrodes for lithium-ion batteries.
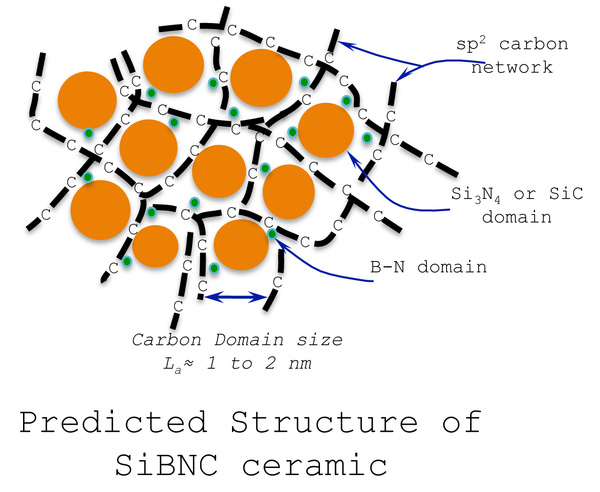
“the ceramic derived from this polymer has a random structure that is generally not observed in traditional ceramics,” the article added. “the silicon in the ceramic bonds to nitrogen and carbon but not boron; boron bonds to nitrogen but not carbon; and carbon bonds to another carbon to form graphene-like strings. this unique structure provides stability at high temperature by delaying reaction with oxygen.”
the patent information can be read at http://patft.uspto.gov/netacgi/nph-parser?sect1=pto1§2=hitoff&d=pall&p=1&u=%2fnetahtml%2fpto%2fsrchnum.htm&r=1
&f=g&l=50&s1=9453111.pn.&os=pn/9453111&rs=pn/9453111.
|